This page contains the detailed and easy notes for AQA GCSE Chemistry Bonding, Structure and Properties of Matter for revision and understanding Bonding, Structure and Properties of Matter.
Banner 1
AQA GCSE Paper 1: Complete Revision Summary
Bonding, Structure and Properties of Matter
4.2 Bonding, Structure and Properties of Matter
- Ionic Bonding
- Covalent Bonding
- Metallic Bonding
- State of Matter
- Ionic compounds
- Covalent Compounds
- Diamond and Graphite
- Nanoparticles
- Graphene and Fullerene
Banner 2
BONDING
Atoms bond to gain full outer shell or noble gas electronic configuration
Ionic Bonding
- transfer of electron between metals and non metals
- between metal and non metals
Covalent Bonding
- sharing of electron between non metals
- between non metals
Metallic Bonding
- Electrostatic force of attraction between fixed positive ions and delocalised electrons.
- between Metals
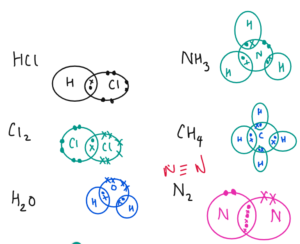
Carbon dioxide (CO2) – Covalent
Ammonia (NH3) – Covalent
Nitrogen (N2) – Covalent
Water (H2O) – Covalent
Sodium chloride -Ionic Bonding
Calcium fluoride— Ionic Bonding
IONIC BONDING: Metals and Non Metals
- It is between a metal and a non metal
- Metal loses an electron and become positively charged.
- Non- Metal gains an electron and becomes negatively charged.
- There is a strong electrostatic force of attraction between opposite charged ions resulting in ionic bonding.
Dot and Cross Diagram
- Write the symbols
- Write electronic configuration
- show outer electrons
- show transfer
- show charges
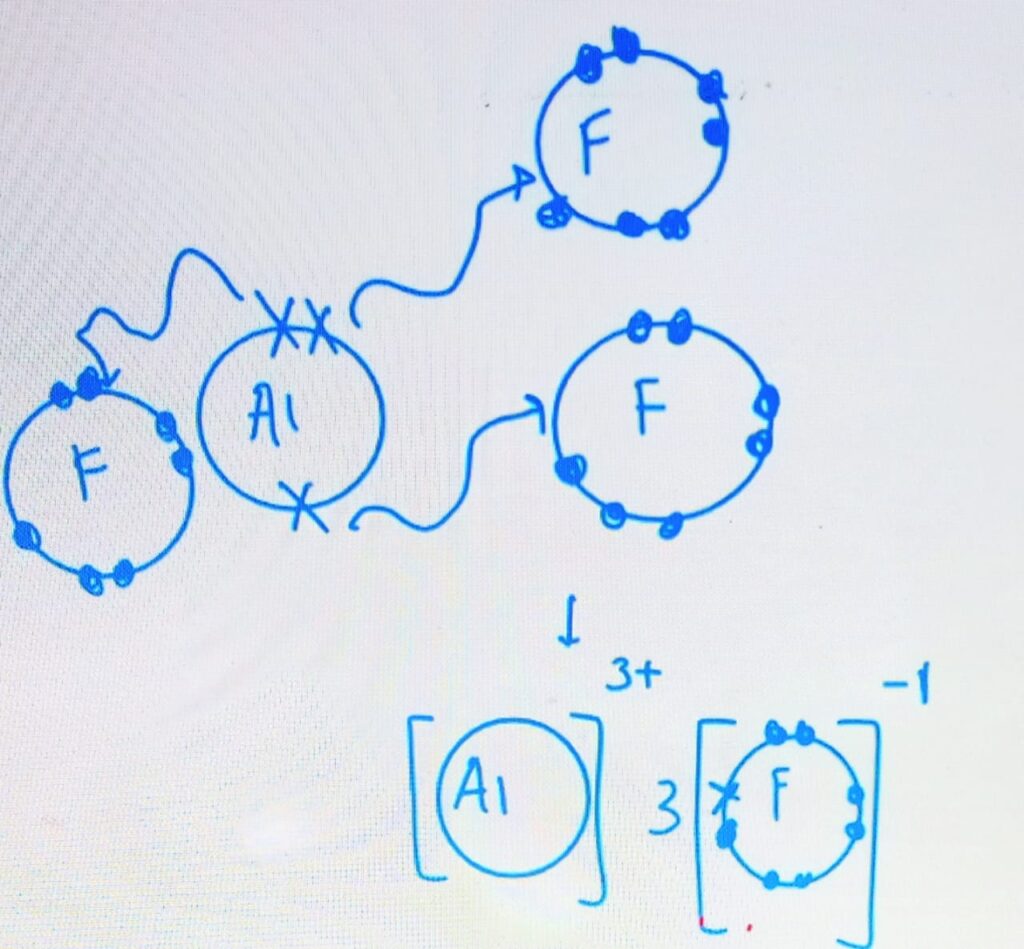
Example – Aluminium Fluoride
Al = 2,8,3
F = 2,7
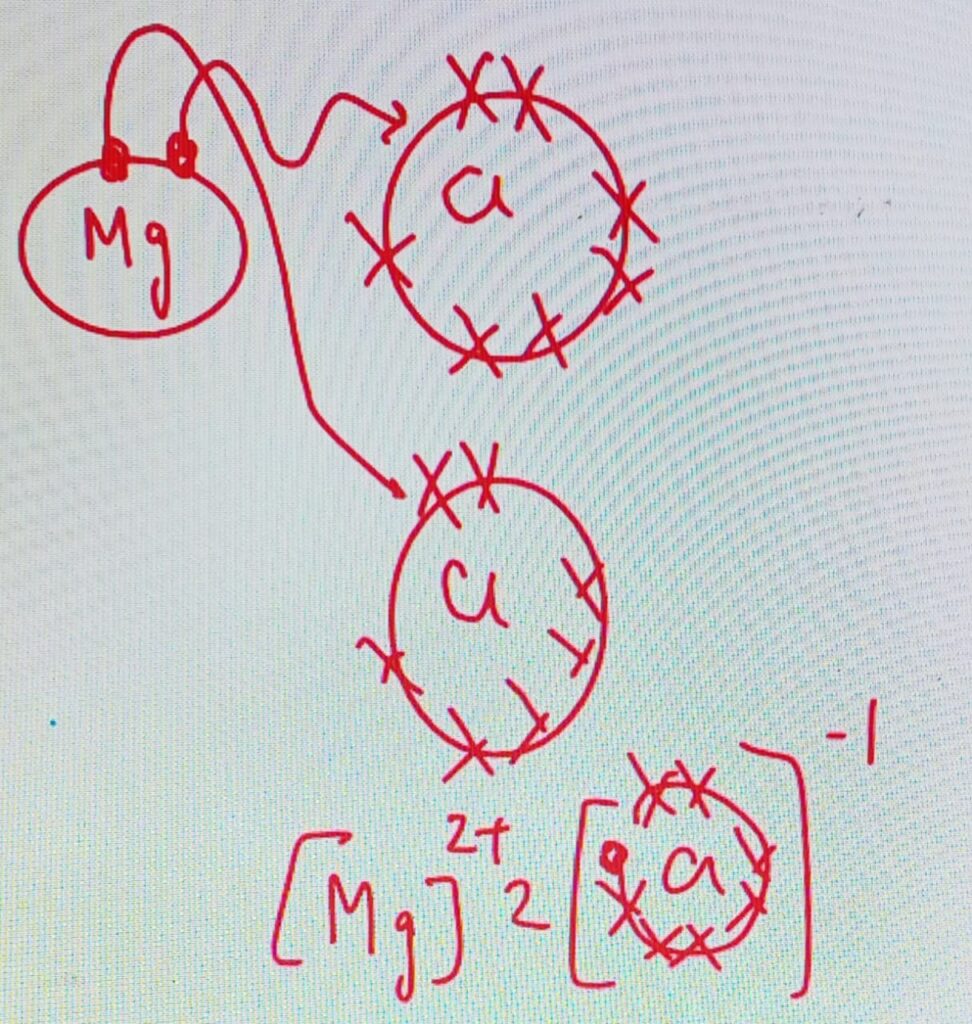
Example – Magnesium chloride
Mg = 2,8,2
Cl = 2,8,7
PROPERTIES OF IONIC COMPOUNDS
Ionic Compound Properties
- Brittle solids with definite crystal shapes
In Ionic compounds, there is a strong electrostatic force of attraction between the opposite charged ions. This results in the formation of giant ionic lattice.
- Good insulators in solid form, but become good conductors in liquid or dissolved form.
In the solid form, the ions are not free to move as they are held together by strong electrostatic force of attraction. In molten or when they are dissolved in water the ions are free to move and conduct electricity.
- High melting and boiling point compared to molecular compounds
In Ionic compounds, there is a strong electrostatic force of attraction between the opposite charged ions. This results in the formation of giant ionic lattice. Large amount of energy is required to overcome the strong electrostatic force of attraction. Therefore, ionic compounds have high melting and boiling point.
Greater the charge of an ionic lattice, stronger is the electrostatic force of attraction. Greater the melting and bp.
For éx – Aluminium chloride > Magnesium chloride > sodium chloride
FORMULAE OF IONIC COMPOUNDS
- Write the Symbols
- Write the charges
- (Upto group the charge is same as the group number. After group 4 it is group number -8)
- Criss Cross
- a) Sodium Oxide = Na2O
- b) Magnesium Nitride = Mg3N2
- c) Calcium Oxide = CaO
- d) Sodium Sulphide = N2S
- e) Sodium Chloride = NaCl
- f) Magnesium chloride = MgCl2
- g) Aluminium Chloride = ACl3


FORMULAE OF COMPLEX IONS
| a) Magnesium Hydroxide | Al2(SO4)3 |
| b) Aluminium Sulphate | Al2(SO4)3 |
| c) Calcium Phosphate | Ca3(PO4)2 |
| d) Magnesium nitrate | Mg(NO3)2 |
| e) Calcium Carbonate | CaCO3 |
| f) Sodium Phosphate | Na3PO4 |
| Positive | Negative |
| Ammonium = NH4+ | Carbonate = CO32- |
| Sulphate = SO42- | |
| Nitrate = NO3– | |
| Phosphate = PO43- | |
| Hydroxide = OH– |
COVALENT BONDING
- It is between two non metals
- It involves the sharing of electrons between two non metals.
- More than one electron pair can also be shared resulting in the formation of single double and triple bonds.
Bonding Structure and properties
Restricted download
×
Properties of Covalent Compounds

Simple Molecule
- Eg – O2, CH4
- They have weak intermolecular forces in them so have a lower melting and a boiling points
- The intermolecular forces increases with increase in size as the surface area between the molecules increases.
- Therefore, polymers which have covalent bonding between them have high melting and boiling point due to increase in chain length.
Giant Covalent
- Diamond
- Graphite
- Silicon Dioxide
GIANT COVALENT STRUCTURES
Substances which have huge network of atoms joined together by covalent bonds form giant covalent structures.


DIAMOND GRAPHITE It is hard. It is soft and greasy. It is an insulator It is a conductor It has a high density. It has a lower density than diamond. Each carbon atom is covalently bonded to four other carbon atoms giving it a strong rigid structure Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized No delocalised electrons present It has delocalised electrons Used in cutting or jewellery It is used in pencil leads.
PROPERTIES OF GRAPHITE
Q1 Why graphite is soft and slippery?
Banner 5
In graphite, Carbon atoms are bonded in the form of layers in the form of hexagons. No covalent bonding between the layers so they can slide past each other. The layers have only weak intermolecular forces between them. By applying a little pressure then layers can easily slide past each other making Graphite soft and slippery.

Q2 Why graphite conduct electricity ?
In graphite, Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized. These delocalized electrons are mobiles electrons which can move and conduct electricity.
FULLERENE AND GRAPHENE
Fullerene: Hollow shaped molecule having hexagonal rings like a bucky ball.
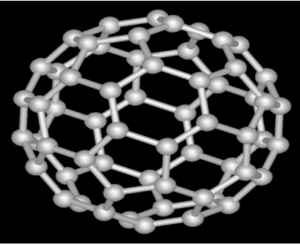
- Also known as bucky ball or buckminsterfullerene.
- Carbon can be in the form of pentagon or hexagon rings
- Used as catalyst, drug delivery and treating cancer.
- Graphene: Layer of interlocking hexagonal rings like single sheet of graphite.
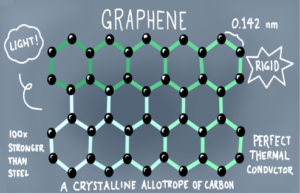
- It is a better conductor than graphite, light and have low density.
- Used in making computer chips and flexible electronic displays.
Baneer 6
CARBON NANOTUBES
- Cylinderical fullerene with the length greater than the diameter.
- High tensile strength – Used in making reinforced composite materials
- High electronic conductivity – used in electronic industry

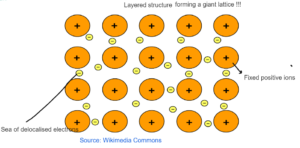
METALLIC BONDING
- It is between two metals.
- There are fixed positive ions present in the sea of delocalised electrons.
- There is strong electrostatic force of attraction between fixed positive ions and delocalized electrons resulting in metallic bonding.
Banner 8
Properties of Metals
Metals are malleable
- Malleable means that the metals can be hammered into any shape.
- Metals have layered structure and layers can slide past each other by hammering giving metals different shapes.
Metals are ductile
- Ductile means that the metals can be drawn into thin wires.
- Metals have layered structure and layers can slide past each other by hammering giving metals a wire shape.
Metallic Bonding
- Atoms in a metal are arranged in a regular manner and vibrate about fixed positions.
- The outermost electrons move freely, forming a ‘sea of electrons’ enveloping the positive metal ions.
Metals are good conductors of electricity
- Metals have delocalised electrons.
- They are mobile and conduct electricity.
- These mobile electrons or delocalised electrons conduct heat and electricity.
Metals have high melting and boiling points
- There is strong electrostatic force of attraction between fixed positive ions and delocalized electrons.
- Large amount of energy is required to overcome strong electrostatic force of attraction.
Banner 9
ALLOYS
- Alloys are the mixture of metals with another metal or a non metal which make the metal stronger.
- In metals the particles are arrranged in layers. There is a regular arrangement of fixed positive ions which can slide past each by applying pressure.
- In alloys there is a mixture of metals with another metal or a non metals. Another metal being different in shape and size distort the regular arrangement of the metal lattice.
- As a result the layers of the metal can no longer slide past each other making it strong
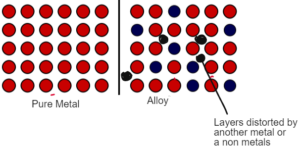
Example: Steel is the alloy of iron which is more strong and resistant to corrossion.
NANOPARTICLES
Nanoparticles are the particles that deals with the paricles of size 1 to 100 nm.
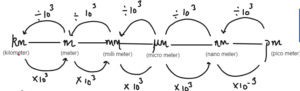
KIL- Killing
MET – Metal
MIL -Milo
MIC – Mickey
NAN – Nano
PIC – Pictures
Convert 10 nm to
- Metre = 10/109m = 10-8m
- Micrometer = 10/106m = 10-5m
SURFACE AREA TO VOLUME RATIO
As the size decreases the surface area to volume ratio Increases.
Therefore Nano particles being very small in size have large surface area to volume ratio making them very useful in Science and Medicine.
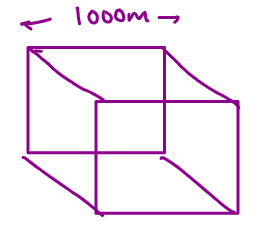
Surface area = 6 x side x side m2
= 6 x 1000 x 1000
= 6 106 m2
Volume = side x side x side
= 109m3
SA: Volume = 6 x 106/109 = 6 x 103 m
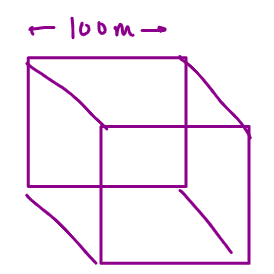
Surface area = 6 x side x side m2
= 6 x 1000 x 1000
= 6 104 m2
Volume = side x side x side
= 106m3
SA: Volume = 6 x 102 m
Banner 10
APPLICATIONS OF NANOPARTICLES
MEDICINES
- To kill cancer and tuomour cells
- For drug delievery
CATALYST
- They have large surface area to volume ratio.
- Used in small quantities so highly effective
COSMETICS
- Used in Sunscreen to block sunlight
HOUSEHOLD
- Self cleaning window panes
- Nano particles breaks dirty in the presence of sunshine which is washed away by water while raining.
RISKS OF NANOPARTICLES
- Due to small size can cause difficulty in breathing
- They can accumulate in the envrionment and cause air pollution
- Due to their large surface area a small spark can result in violent explosion making them risky to use.
- They are toxic and cause breathing and respiratory problems.
- Due to their small size they can also cause water pollution and risk the aquation life.
STATES OF MATTER
SOLIDS LIQUIDS GASES Particles are close to each other. Particles are slightly closer to each other. Particles are far apart. Have fixed shape Do not have fixed shape Do not have fixed shape Strong forces between the particles Weak forces between the particles Very weak forces between the particles. Have definite volume Have fixed volume Do not have fixed volume cannot be compressed Can be compressed Highly compressible Cannot flow Can flow Can flow
Banner 11
KEY TERMS !!!!
Ions – charged atoms with unequal number of protons and electrons
Ionic Bonding — bond formed between a metal and a non metal which involves complete transfer of electrons from metal to a non metal
Dot and Cross — diagram that show transfer of electron in an ionic bond or sharing of electrons in a covalent bond.
Covalent Bonding— bonding between two non metals which involves sharing of electrons.
Metallic Bonding— bonding in metals which involves strong electrostatic forces of attraction between fixed positive ions and delocalised electrons.
Intermolecular Forces — The forces between the molecules which determines the melting or a boiling point.
Giant Covalent Molecules — Covalently bonded molecules which forms large giant structure
Polymers – Molecules which are made up of many repeating units
Delocalised electrons — Mobile electrons that are free to move as they are not associated with a bond or an atom.
Fullerene— Allotrope of carbon which forms a cage like structure like bucky ball.
Graphene— Allotrope of carbon which is equivalent to single layer of graphite
Alloys— Mixture of metals with another metal or a non metal.
Nanoparticles- particles which are of the size of 1 nm to 100 nm_
Nanoscience—lt is the branch of science that deals with nanoparticles
State Of Matter-Different forms that a matter can take They are solids, liquids and gas
Solids — States of matter with fixed shape and volume.
Liquids— States of matter without fixed shape but fixed volume.
Gases— States of matter with fixed shape and volume.
TEST YOURSELF
Q1 Name the type of bonding in the following compounds :
- a) Sodium Chloride – Ionic
- b) Magnesium – Metallic
- c) Nitrogen – Covalent
- d) Carbon Dioxide – Covalent
- e) Water – Covalent
- f) Ammonia – Covalent
Q2 Draw dot and cross diagram to represent bonding in the following
- a) Sodium chloride
- b) Water
- c) Magnesium
Q3 Differentiate Between Diamond and Graphite
DIAMOND GRAPHITE It is hard. It is soft and greasy. It is an insulator It is a conductor It has a high density. It has a lower density than diamond. Each carbon atom is covalently bonded to four other carbon atoms giving it a strong rigid structure Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized No delocalised electrons present It has delocalised electrons Used in cutting or jewellery It is used in pencil leads.
Q4 Why Ionic compounds do not conduct electricity in solids ?
In solids, the ions are held together by strong electrostatic force of attraction in the giant ionic lattice. In molten state the ions are free to move therefore conduct electricity
Q5 Why Alloys are stronger than metals
Alloys are the mixture of metals which distors the regular arrangement of metal as a result of which layers are not able to slide past each other making alloys stronger than metals.
Q6 Why alumunium has a stronger melting point than sodium
Aluminium has a greater charge. Due to greater charge of aluminium there is a stronger electrostatic forces of attraction between fixed positive ions and delocalised elecctrons. As a result aluminium has a greater melting point than sodium.
Q7 What are nanoparticles? Write the properties and applications of nanoparticles
Nanoparticles are the particles between the size of 1 to 100 nm_ Due to smaller size they have large surface area to volume ratio making them highly useful in medicine, catalysts, cosmetics and electronic industry.
Banner 12
Disclaimer:
I have tried my level best to cover the maximum of your specification. But this is not the alternative to the textbook. You should cover the specification or the textbook thoroughly. This is the quick revision to help you cover the gist of everything. In case you spot any errors then do let us know and we will rectify it.
References:
BBC Bitesize
Wikipedia
Wikimedia Commons
Image Source:
Wikipedia
Wikimedia
Commons
Flickr
Pixabay
Questions by Topic
Make sure you have watched the above videos and are familiar with the key definations before trying these questions. It is also good to time yourself while doing these questions so that you can work on the speed as well.
C3- Structure And Bonding
- Bonding & Structuce 1 MS
- Bonding & Structuce 1 QP
- Bonding & Structuce 2 QP
- Bonding & Structuce 2 MS
- Bonding & Structuce 3 QP
- Bonding & Structuce 3 MS
- Bulk & Surface Properties of Matter 1 QP
- Bulk & Surface Properties of Matter 1 MS
- Bulk & Surface Properties of Matter 2 QP
- Bulk & Surface Properties of Matter 2 MS
- Bulk & Surface Properties of Matter 3 QP
- Bulk & Surface Properties of Matter 3 MS
- Ionic, Covalent & Metallic Bonds 1 QP
- Ionic, Covalent & Metallic Bonds 1 MS
- Ionic, Covalent & Metallic Bonds 2 QP
- Ionic, Covalent & Metallic Bonds 2 MS
- Ionic, Covalent & Metallic Bonds 3 QP
- Ionic, Covalent & Metallic Bonds 3 MS
- Structure & Bonding Carbon 1 QP
- Structure & Bonding Carbon 1 MS
- Structure & Bonding Carbon 2 QP
- Structure & Bonding Carbon 2 MS
- Structure & Bonding Carbon 3 QP
- Structure & Bonding Carbon 3 MS
Restricted download
×Properties of Covalent Compounds

Simple Molecule
- Eg – O2, CH4
- They have weak intermolecular forces in them so have a lower melting and a boiling points
- The intermolecular forces increases with increase in size as the surface area between the molecules increases.
- Therefore, polymers which have covalent bonding between them have high melting and boiling point due to increase in chain length.
Giant Covalent
- Diamond
- Graphite
- Silicon Dioxide
GIANT COVALENT STRUCTURES
Substances which have huge network of atoms joined together by covalent bonds form giant covalent structures.


| DIAMOND | GRAPHITE |
| It is hard. | It is soft and greasy. |
| It is an insulator | It is a conductor |
| It has a high density. | It has a lower density than diamond. |
| Each carbon atom is covalently bonded to four other carbon atoms giving it a strong rigid structure | Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized |
| No delocalised electrons present | It has delocalised electrons |
| Used in cutting or jewellery | It is used in pencil leads. |
PROPERTIES OF GRAPHITE
Q1 Why graphite is soft and slippery?
Banner 5
In graphite, Carbon atoms are bonded in the form of layers in the form of hexagons. No covalent bonding between the layers so they can slide past each other. The layers have only weak intermolecular forces between them. By applying a little pressure then layers can easily slide past each other making Graphite soft and slippery.

Q2 Why graphite conduct electricity ?
In graphite, Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized. These delocalized electrons are mobiles electrons which can move and conduct electricity.
FULLERENE AND GRAPHENE
Fullerene: Hollow shaped molecule having hexagonal rings like a bucky ball.

- Also known as bucky ball or buckminsterfullerene.
- Carbon can be in the form of pentagon or hexagon rings
- Used as catalyst, drug delivery and treating cancer.
- Graphene: Layer of interlocking hexagonal rings like single sheet of graphite.

- It is a better conductor than graphite, light and have low density.
- Used in making computer chips and flexible electronic displays.
Baneer 6
CARBON NANOTUBES
- Cylinderical fullerene with the length greater than the diameter.
- High tensile strength – Used in making reinforced composite materials
- High electronic conductivity – used in electronic industry

METALLIC BONDING
- It is between two metals.
- There are fixed positive ions present in the sea of delocalised electrons.
- There is strong electrostatic force of attraction between fixed positive ions and delocalized electrons resulting in metallic bonding.

Banner 8
Properties of Metals
Metals are malleable
- Malleable means that the metals can be hammered into any shape.
- Metals have layered structure and layers can slide past each other by hammering giving metals different shapes.
Metals are ductile
- Ductile means that the metals can be drawn into thin wires.
- Metals have layered structure and layers can slide past each other by hammering giving metals a wire shape.
Metallic Bonding
- Atoms in a metal are arranged in a regular manner and vibrate about fixed positions.
- The outermost electrons move freely, forming a ‘sea of electrons’ enveloping the positive metal ions.
Metals are good conductors of electricity
- Metals have delocalised electrons.
- They are mobile and conduct electricity.
- These mobile electrons or delocalised electrons conduct heat and electricity.
Metals have high melting and boiling points
- There is strong electrostatic force of attraction between fixed positive ions and delocalized electrons.
- Large amount of energy is required to overcome strong electrostatic force of attraction.
Banner 9
ALLOYS
- Alloys are the mixture of metals with another metal or a non metal which make the metal stronger.
- In metals the particles are arrranged in layers. There is a regular arrangement of fixed positive ions which can slide past each by applying pressure.
- In alloys there is a mixture of metals with another metal or a non metals. Another metal being different in shape and size distort the regular arrangement of the metal lattice.
- As a result the layers of the metal can no longer slide past each other making it strong

Example: Steel is the alloy of iron which is more strong and resistant to corrossion.
NANOPARTICLES
Nanoparticles are the particles that deals with the paricles of size 1 to 100 nm.

KIL- Killing
MET – Metal
MIL -Milo
MIC – Mickey
NAN – Nano
PIC – Pictures
Convert 10 nm to
- Metre = 10/109m = 10-8m
- Micrometer = 10/106m = 10-5m
SURFACE AREA TO VOLUME RATIO
As the size decreases the surface area to volume ratio Increases.
Therefore Nano particles being very small in size have large surface area to volume ratio making them very useful in Science and Medicine.

Surface area = 6 x side x side m2
= 6 x 1000 x 1000
= 6 106 m2
Volume = side x side x side
= 109m3
SA: Volume = 6 x 106/109 = 6 x 103 m

Surface area = 6 x side x side m2
= 6 x 1000 x 1000
= 6 104 m2
Volume = side x side x side
= 106m3
SA: Volume = 6 x 102 m
Banner 10
APPLICATIONS OF NANOPARTICLES
MEDICINES
- To kill cancer and tuomour cells
- For drug delievery
CATALYST
- They have large surface area to volume ratio.
- Used in small quantities so highly effective
COSMETICS
- Used in Sunscreen to block sunlight
HOUSEHOLD
- Self cleaning window panes
- Nano particles breaks dirty in the presence of sunshine which is washed away by water while raining.
RISKS OF NANOPARTICLES
- Due to small size can cause difficulty in breathing
- They can accumulate in the envrionment and cause air pollution
- Due to their large surface area a small spark can result in violent explosion making them risky to use.
- They are toxic and cause breathing and respiratory problems.
- Due to their small size they can also cause water pollution and risk the aquation life.
STATES OF MATTER
| SOLIDS | LIQUIDS | GASES |
| Particles are close to each other. | Particles are slightly closer to each other. | Particles are far apart. |
| Have fixed shape | Do not have fixed shape | Do not have fixed shape |
| Strong forces between the particles | Weak forces between the particles | Very weak forces between the particles. |
| Have definite volume | Have fixed volume | Do not have fixed volume |
| cannot be compressed | Can be compressed | Highly compressible |
| Cannot flow | Can flow | Can flow |
Banner 11
KEY TERMS !!!!
Ions – charged atoms with unequal number of protons and electrons
Ionic Bonding — bond formed between a metal and a non metal which involves complete transfer of electrons from metal to a non metal
Dot and Cross — diagram that show transfer of electron in an ionic bond or sharing of electrons in a covalent bond.
Covalent Bonding— bonding between two non metals which involves sharing of electrons.
Metallic Bonding— bonding in metals which involves strong electrostatic forces of attraction between fixed positive ions and delocalised electrons.
Intermolecular Forces — The forces between the molecules which determines the melting or a boiling point.
Giant Covalent Molecules — Covalently bonded molecules which forms large giant structure
Polymers – Molecules which are made up of many repeating units
Delocalised electrons — Mobile electrons that are free to move as they are not associated with a bond or an atom.
Fullerene— Allotrope of carbon which forms a cage like structure like bucky ball.
Graphene— Allotrope of carbon which is equivalent to single layer of graphite
Alloys— Mixture of metals with another metal or a non metal.
Nanoparticles- particles which are of the size of 1 nm to 100 nm_
Nanoscience—lt is the branch of science that deals with nanoparticles
State Of Matter-Different forms that a matter can take They are solids, liquids and gas
Solids — States of matter with fixed shape and volume.
Liquids— States of matter without fixed shape but fixed volume.
Gases— States of matter with fixed shape and volume.
TEST YOURSELF
Q1 Name the type of bonding in the following compounds :
- a) Sodium Chloride – Ionic
- b) Magnesium – Metallic
- c) Nitrogen – Covalent
- d) Carbon Dioxide – Covalent
- e) Water – Covalent
- f) Ammonia – Covalent
Q2 Draw dot and cross diagram to represent bonding in the following
- a) Sodium chloride
- b) Water
- c) Magnesium
Q3 Differentiate Between Diamond and Graphite
| DIAMOND | GRAPHITE |
| It is hard. | It is soft and greasy. |
| It is an insulator | It is a conductor |
| It has a high density. | It has a lower density than diamond. |
| Each carbon atom is covalently bonded to four other carbon atoms giving it a strong rigid structure | Carbon atoms are bonded in the form of layer in the form of hexagons. No covalent bonding between the layers so they can slide past. Each carbon atom is bonded with three other carbon leaving the fourth electron has delocalized |
| No delocalised electrons present | It has delocalised electrons |
| Used in cutting or jewellery | It is used in pencil leads. |
Q4 Why Ionic compounds do not conduct electricity in solids ?
In solids, the ions are held together by strong electrostatic force of attraction in the giant ionic lattice. In molten state the ions are free to move therefore conduct electricity
Q5 Why Alloys are stronger than metals
Alloys are the mixture of metals which distors the regular arrangement of metal as a result of which layers are not able to slide past each other making alloys stronger than metals.
Q6 Why alumunium has a stronger melting point than sodium
Aluminium has a greater charge. Due to greater charge of aluminium there is a stronger electrostatic forces of attraction between fixed positive ions and delocalised elecctrons. As a result aluminium has a greater melting point than sodium.
Q7 What are nanoparticles? Write the properties and applications of nanoparticles
Nanoparticles are the particles between the size of 1 to 100 nm_ Due to smaller size they have large surface area to volume ratio making them highly useful in medicine, catalysts, cosmetics and electronic industry.
Banner 12
Disclaimer:
I have tried my level best to cover the maximum of your specification. But this is not the alternative to the textbook. You should cover the specification or the textbook thoroughly. This is the quick revision to help you cover the gist of everything. In case you spot any errors then do let us know and we will rectify it.
References:
BBC Bitesize
Wikipedia
Wikimedia Commons
Image Source:
Wikipedia
Wikimedia
Commons
Flickr
Pixabay
Make sure you have watched the above videos and are familiar with the key definations before trying these questions. It is also good to time yourself while doing these questions so that you can work on the speed as well.
C3- Structure And Bonding
- Bonding & Structuce 1 MS
- Bonding & Structuce 1 QP
- Bonding & Structuce 2 QP
- Bonding & Structuce 2 MS
- Bonding & Structuce 3 QP
- Bonding & Structuce 3 MS
- Bulk & Surface Properties of Matter 1 QP
- Bulk & Surface Properties of Matter 1 MS
- Bulk & Surface Properties of Matter 2 QP
- Bulk & Surface Properties of Matter 2 MS
- Bulk & Surface Properties of Matter 3 QP
- Bulk & Surface Properties of Matter 3 MS
- Ionic, Covalent & Metallic Bonds 1 QP
- Ionic, Covalent & Metallic Bonds 1 MS
- Ionic, Covalent & Metallic Bonds 2 QP
- Ionic, Covalent & Metallic Bonds 2 MS
- Ionic, Covalent & Metallic Bonds 3 QP
- Ionic, Covalent & Metallic Bonds 3 MS
- Structure & Bonding Carbon 1 QP
- Structure & Bonding Carbon 1 MS
- Structure & Bonding Carbon 2 QP
- Structure & Bonding Carbon 2 MS
- Structure & Bonding Carbon 3 QP
- Structure & Bonding Carbon 3 MS
