Notes
Question by Topic
Notes
This page contains the detailed and easy notes for GCSE Edexcel Groups in the Periodic Table for revision and understanding Groups in the Periodic Table.
Banner 1GCSE Edexcel Complete Revision Summary
Groups in the Periodic Table
Groups in the periodic table
- The periodic table
- Development of the periodic table
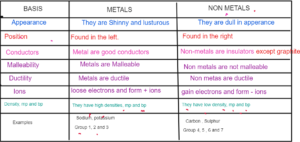
- Metals and non-metals
- Group 0
- Group 1
- Group 7

- Argon atoms have a greater relative mass than potassium which will place Argon in the group of sodium and lithium and potassium in the group of noble gases.
- Many other elements were found not fitting this pattern and were swapped by Mendleev’s to maintain the periodicity.
- Organise the elements in the order of increasing atomic number
- All the shortcoming due to atomic weights were solved by organising the elements in the order of increasing atomic number.
- Heavy atoms are due to the presence of different isotopes of the elements.
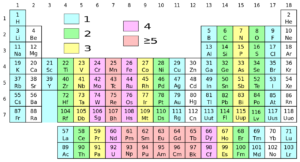
Groups in the Periodic Table
Group 1: Alkali Metals
 GROUP 1: Alkali Metals Physical Properties
GROUP 1: Alkali Metals Physical Properties
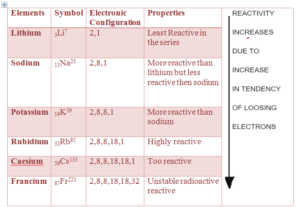
- They are highly reactive
- Reactivity increases down the group – because tendency to loose one electron increases down the group due to increase increase in size and decrease in nuclear charge
- They loose one electron and form +1 Ions.
- They are stored in kerosene or oil to prevent them reacting from air and water
- They are soft, silvery and shinny.
- They look dull in air as they react with oxygen and form oxide which coats their surface
- Lithium is less reactive and francium is highly reactive.
- They have low melting and boiling point and the melting and boiling point decreases down the group.
Groups in the Periodic Table (1)
Restricted download
×
Group 1: Alkali Metals Chemical Properties
REACTION WITH WATER
Reacts with water to metal hydroxide
2M + H2O 2MOH + H2
Metal hydroxide are alkali therefore the pH increases. Reactivity increases down the group so potassium reacts violently
Fizzing is produced due to the formation of hydrogen.
Eg – 2Li(s) + H2O(l) 2LiOH(aq) + H2(g)
Reaction with Oxygen
Reacts with oxygen to form a metal oxide
2M + O2 M2O
Metals go dull in air due to this reaction
4Li(s) + 4O2(g) 2Li2O(s)
Reaction with Halogens
React with halogens to form metal Halides
2M +X2 2MX [X= F, Cl, Br, I]
Metal Halides are while solids but dissolve in water to form colourless solutions.
2Li(s) + F2(g) 2LiF(s)
LITHIUM
SODIUM
POTASSIUM
2Li(s) + 2H2O (l) 2LiOH(aq) + H2(g)
2Na(s) + 2H2O (l) 2NaOH(aq) + H2(g)
2K(s) + H2O (l) KOH(aq) + H2(g)
Floats in water due to less denisty than water
Floats in water due to less density than water
Floats in water due to less density than water
Fizzes due to the formation of hydrogen gas.
Fizzes due to the formation of hydrogen gas.
Fizzes due to the formation of hydrogen gas.
Shape is retained while reacting and gets smaller.
It melts into a call while reacting.
Melts into a ball, catches fire and produces a lilac flame.
Banner 3
WHY REACTIVITY OF GROUP 1 INCREASES DOWN THE GROUP ?
The Reactivity of Group 1 increases down the group as the tendency to loose an electron increases down the group. – React by loosing an electron
FACTORS AFFECTING TENDENCY TO LOOSE AN ELECTRON
- To loose an electron small nuclear charge greater size of atom and greater shielding is required
- Nuclear charge – Great the size of the atom, the outer electron becomes further away from the nucleus decreasing the nuclear charge
- Shielding – More the number of inner electrons due to increases in number of shell greater will be the shielding of the outer electron from the nuclear charge
- Size of the atoms – Greater the size of the atom, the outer electron will become further away from the nucleus resulting in decreases in nuclear charge
- Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus.
- As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge.
- Therefore, the tendency of atom to loose an electron increases down the grou resulting in increase in reactivity down the group.
GROUP 7 : Halogens ( Salt Forming)
Element
Symbol
Electronic Configuration
State at Room Temperature
Florine
9F19
2,7
Yellow Gas
Chlorine
17Cl35
2,8,7
Green Gas and Pale green in solution
Bromine
35Br80
2,8,18,7
Volatile Brown Liquid –yellow in solution
Iodine
53I127
2,8,18,18,7
Volatile Purple solid – Brown in Solution
Astatine
85At210
2,8,18,32,18,7
Radioactive
Banner 4
GROUP 7 : Halogens Physical Properties
- They are non metals
- They gain an electron to form -1 ions.
- They have low melting and boiling points
- Their melting point increases down the group due to increases in intermolecular forces.
- They are found in pairs and exist as diatomic molecules (X2)
- They are poisonous and smelly
- Their reactivity increase down the group
- Their density increases down the group.
- They are poor conductors of heat and electricity
HALOGEN REACTION X[F,Cl,Br,I]
REACTION WITH HYDROGEN
They react with hydrogen to form hydrogen halides.
X2(s) + H2(g) 2HX(g)
Reactivity decreases down the group so fluorine and chlorine reacts explosively and bromine and iodine reacts at higher temperature in the presence of catalyst.
REACTION WITH METALS
They react with metals to form ionic compounds. In Ionic compounds, halogens gain one electron from the metals to form -1 ions and attain noble gas electronic configurations.
2Na(s) + Cl2(g) NaCl(s)
Mg(s) + Cl2(g) MgCl2(s)
DISPLACEMENT REACTION
The more reactive halogen displaces the less reactive halogen from its salt
As the reactivity decreases down the group, the halogen at the top can take the position of the halogen at the bottom in its compounds and will displace the less reactive halogen.
Cl2 + 2NaBr 2NaCl + Br2(yellow solution formed)
(more (salt of less reactive (chlorine being more reactive has taken the reactive) halogen than Chlorine) position of less reactive bromine in its
compounds)
Cl2 +2NaF No reaction
F2 – can displace all halogens
Cl2 – can displace all Halogen except Fluorine
Br2 – can displace only Iodine
F Cl Br I
decreasing reactivity
WHY REACTIVITY OF GROUP 7 DECREASES DOWN THE GROUP ?
The Reactivity of Group 7 decreases down the group as the electron affinity or tendency to gain the electron decreases down the qroup. – React by gaining electron
Banner 5
FACTORS AFFECTING TENDENCY TO GAIN AN ELECTRON
- To gain an electron, smaller nuclear charge smaller size and less shielding is required
- Nuclear Charge – Smaller the size of the atom, greater will be the force of the nucleus as the electron will be closer to the nucleus.
- Shielding – Less electrons and shells, smaller will *be the shielding which will in turn increa the nuclear charge.
- Size of Atom – Greater the size of the atom, the outer electron will become further away from th nucleus resulting in decreases in nuclear charge
- Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases.
- The size of the atom also increases down the group which makes the nuclear charge weaker
- The electron shells also increases which decreases the effective nuclear charge on the incoming electron.
- Due to all these factors, the nuclear charge decreases which decreases the tendency of gaining electrons down the group of halogen making them less reactive.
COMPARISON BETWEEN GROUP 1 and GROUP 7
GROUP 1 Alkali Metals
GROUP 7 Halogens
a) Have one electron in their outermost shells
Have seven electrons in their outermost shell
b) They are metals
They are non metals
c) They react by loosing electrons
They react by gaining electrons
d) They form +1 ions
They form -1 ions
e) Their reactivitiy increases down the group
Their reactivity decreases down the group
f) Reactivity depends on tendency to loose an electron
Reactivity depends on tendency to gain an electron
Baneer 6
TRANSITION METALS
- Found between group 2 and group 3
- They are hard
- They are strong
- They are malleable and ductile
- They have higher densities than group 1 and group 2 hence they are used in construction purpose like iron.
- They show variable oxidation states
- They are used commercially as catalyst
- They form coloured compounds.
- They show the reaction with oxygen, water and halogen like group 1 but they react much slowly than alkali metals.
 Banner 7
BASIC TERMS OF PERIODIC TABLE
Periodic Table – A table that shows arrangement of all the known elements in the order of increasing atomic number. The table is organised into periods and groups.
Metals- Elements found to the left of the periodic table which are soft, shinny, conductors malleable and ductile eg Group 1; group 2 and group 3 elements
Non Metals — Elements found to the right of the periodic table which are dull, insulators Group 4,516 and 7 are non metals.
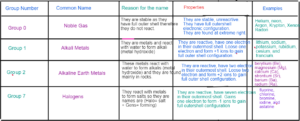
Halogens Group 7 elements are halogens as they are salt forming.
Alkali Metals Group 1 elements which react with water to form alkali
Noble Gases — Group 0 elements which are stable and do not react as they have complete outer shell
Elements found between group 2 and group 3 which have high densities, show variable oxidation states
Transition Metals and form coloured compounds
Periods- Horizontal rows of the periodic table
Groups – Vertical columns of the periodic table
Group Number – Indicates the number of electrons in the outermost shell.
Alkali Bases that are soluble in water
Displacement Reaction — When a more reactive element displaces the less reactive element from its salt
Banner 8
Q1 Look at the periodic table and give two examples of each
Metal — Li, Na
Non Metal – O2, F2
Alkali Metal – K, Li
Halogens – F, Cl
Noble Gas – He, Ar
Semi metal or metalloid – Si
Metals that form +1 ions – Li, K
Non metal that form -1 ions – F, Cl
Metal that form +2 ions – Mg, Ca
Transition metal – Fe, Cu
Q2 Write the name of most reactive halogen and most reactive alkali metals
Halogen –F Alkali Metal – Fr
Q3 Why the alkali metals gets more reactive down the group
Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus. As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge. Therefore, the tendency of atom to loose an electron increases down the group resulting in increase in reactivity down the group.
Q4 Why halogens get less reactive down the group
Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases. The size of the atom also increases down the group which makes the nuclear charge weaker. The electron shells also increases which decreases the effective nuclear charge on the incoming electron. Due to all these factors, the nuclear charge decreases which decreeases the tendency of gaining electrons down the group of halogen making them less reactive.
Q5 Write the balanced chemical equation with state symbols of
Banner 7
BASIC TERMS OF PERIODIC TABLE
Periodic Table – A table that shows arrangement of all the known elements in the order of increasing atomic number. The table is organised into periods and groups.
Metals- Elements found to the left of the periodic table which are soft, shinny, conductors malleable and ductile eg Group 1; group 2 and group 3 elements
Non Metals — Elements found to the right of the periodic table which are dull, insulators Group 4,516 and 7 are non metals.
Halogens Group 7 elements are halogens as they are salt forming.
Alkali Metals Group 1 elements which react with water to form alkali
Noble Gases — Group 0 elements which are stable and do not react as they have complete outer shell
Elements found between group 2 and group 3 which have high densities, show variable oxidation states
Transition Metals and form coloured compounds
Periods- Horizontal rows of the periodic table
Groups – Vertical columns of the periodic table
Group Number – Indicates the number of electrons in the outermost shell.
Alkali Bases that are soluble in water
Displacement Reaction — When a more reactive element displaces the less reactive element from its salt
Banner 8
Q1 Look at the periodic table and give two examples of each
Metal — Li, Na
Non Metal – O2, F2
Alkali Metal – K, Li
Halogens – F, Cl
Noble Gas – He, Ar
Semi metal or metalloid – Si
Metals that form +1 ions – Li, K
Non metal that form -1 ions – F, Cl
Metal that form +2 ions – Mg, Ca
Transition metal – Fe, Cu
Q2 Write the name of most reactive halogen and most reactive alkali metals
Halogen –F Alkali Metal – Fr
Q3 Why the alkali metals gets more reactive down the group
Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus. As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge. Therefore, the tendency of atom to loose an electron increases down the group resulting in increase in reactivity down the group.
Q4 Why halogens get less reactive down the group
Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases. The size of the atom also increases down the group which makes the nuclear charge weaker. The electron shells also increases which decreases the effective nuclear charge on the incoming electron. Due to all these factors, the nuclear charge decreases which decreeases the tendency of gaining electrons down the group of halogen making them less reactive.
Q5 Write the balanced chemical equation with state symbols of
- a) Potassium with water
2K(s) + 2H2O(g) 2KOH(aq) +H2(g)
- b) Lithium with oxygen
4Li(s) + O2(g) 2Li2O (s)
- c) Sodium with bromine
2Na(s) + Br2(l) 2NaBr (s)
- d) Chlorine with hydrogen
Cl2 (s) + H2(g) 2HCl (g)
Q6 Explain displacement reaction of halogens with examples.
F2 + 2NaCl 2NaF + Cl2
Cl2 +2NaF No reaction
Banner 9
Disclaimer:
I have tried my level best to cover the maximum of your specification. But this is not the alternative to the textbook. You should cover the specification or the textbook thoroughly. This is the quick revision to help you cover the gist of everything. In case you spot any errors then do let us know and we will rectify it.
References:
BBC Bitesize
Wikipedia
Wikimedia Commons
Image Source:
Wikipedia
Wikimedia
Commons
Flickr
Pixabay
Question by Topic
Make sure you have watched the above videos and are familiar with the key definations before trying these questions. It is also good to time yourself while doing these questions so that you can work on the speed as well.
Chemistry Of The Periodic Table
- Periodic Table 1 MS
- Periodic Table 1 QP
- Periodic Table 2 MS
- Periodic Table 2 QP
- Periodic Table 3 MS
- Periodic Table 3 QP
- Properties of Transition Metals 1 MS
- Properties of Transition Metals 1 QP
- Properties of Transition Metals 2 MS
- Properties of Transition Metals 2 QP
- Properties of Transition Metals 3 MS
- Properties of Transition Metals 3 QP
Restricted download
×Group 1: Alkali Metals Chemical Properties
REACTION WITH WATER Reacts with water to metal hydroxide 2M + H2O 2MOH + H2 Metal hydroxide are alkali therefore the pH increases. Reactivity increases down the group so potassium reacts violently Fizzing is produced due to the formation of hydrogen. Eg – 2Li(s) + H2O(l) 2LiOH(aq) + H2(g)Reaction with Oxygen
Reacts with oxygen to form a metal oxide 2M + O2 M2O Metals go dull in air due to this reaction 4Li(s) + 4O2(g) 2Li2O(s)Reaction with Halogens
React with halogens to form metal Halides 2M +X2 2MX [X= F, Cl, Br, I] Metal Halides are while solids but dissolve in water to form colourless solutions. 2Li(s) + F2(g) 2LiF(s)| LITHIUM | SODIUM | POTASSIUM |
| 2Li(s) + 2H2O (l) 2LiOH(aq) + H2(g) | 2Na(s) + 2H2O (l) 2NaOH(aq) + H2(g) | 2K(s) + H2O (l) KOH(aq) + H2(g) |
| Floats in water due to less denisty than water | Floats in water due to less density than water | Floats in water due to less density than water |
| Fizzes due to the formation of hydrogen gas. | Fizzes due to the formation of hydrogen gas. | Fizzes due to the formation of hydrogen gas. |
| Shape is retained while reacting and gets smaller. | It melts into a call while reacting. | Melts into a ball, catches fire and produces a lilac flame. |
WHY REACTIVITY OF GROUP 1 INCREASES DOWN THE GROUP ?
The Reactivity of Group 1 increases down the group as the tendency to loose an electron increases down the group. – React by loosing an electronFACTORS AFFECTING TENDENCY TO LOOSE AN ELECTRON
- To loose an electron small nuclear charge greater size of atom and greater shielding is required
- Nuclear charge – Great the size of the atom, the outer electron becomes further away from the nucleus decreasing the nuclear charge
- Shielding – More the number of inner electrons due to increases in number of shell greater will be the shielding of the outer electron from the nuclear charge
- Size of the atoms – Greater the size of the atom, the outer electron will become further away from the nucleus resulting in decreases in nuclear charge
- Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus.
- As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge.
- Therefore, the tendency of atom to loose an electron increases down the grou resulting in increase in reactivity down the group.
GROUP 7 : Halogens ( Salt Forming)
| Element | Symbol | Electronic Configuration | State at Room Temperature |
| Florine | 9F19 | 2,7 | Yellow Gas |
| Chlorine | 17Cl35 | 2,8,7 | Green Gas and Pale green in solution |
| Bromine | 35Br80 | 2,8,18,7 | Volatile Brown Liquid –yellow in solution |
| Iodine | 53I127 | 2,8,18,18,7 | Volatile Purple solid – Brown in Solution |
| Astatine | 85At210 | 2,8,18,32,18,7 | Radioactive |
GROUP 7 : Halogens Physical Properties
- They are non metals
- They gain an electron to form -1 ions.
- They have low melting and boiling points
- Their melting point increases down the group due to increases in intermolecular forces.
- They are found in pairs and exist as diatomic molecules (X2)
- They are poisonous and smelly
- Their reactivity increase down the group
- Their density increases down the group.
- They are poor conductors of heat and electricity
HALOGEN REACTION X[F,Cl,Br,I]
REACTION WITH HYDROGEN They react with hydrogen to form hydrogen halides. X2(s) + H2(g) 2HX(g) Reactivity decreases down the group so fluorine and chlorine reacts explosively and bromine and iodine reacts at higher temperature in the presence of catalyst. REACTION WITH METALS They react with metals to form ionic compounds. In Ionic compounds, halogens gain one electron from the metals to form -1 ions and attain noble gas electronic configurations. 2Na(s) + Cl2(g) NaCl(s) Mg(s) + Cl2(g) MgCl2(s) DISPLACEMENT REACTION The more reactive halogen displaces the less reactive halogen from its salt As the reactivity decreases down the group, the halogen at the top can take the position of the halogen at the bottom in its compounds and will displace the less reactive halogen. Cl2 + 2NaBr 2NaCl + Br2(yellow solution formed) (more (salt of less reactive (chlorine being more reactive has taken the reactive) halogen than Chlorine) position of less reactive bromine in its compounds) Cl2 +2NaF No reaction F2 – can displace all halogens Cl2 – can displace all Halogen except Fluorine Br2 – can displace only Iodine F Cl Br I decreasing reactivityWHY REACTIVITY OF GROUP 7 DECREASES DOWN THE GROUP ?
The Reactivity of Group 7 decreases down the group as the electron affinity or tendency to gain the electron decreases down the qroup. – React by gaining electron Banner 5FACTORS AFFECTING TENDENCY TO GAIN AN ELECTRON
- To gain an electron, smaller nuclear charge smaller size and less shielding is required
- Nuclear Charge – Smaller the size of the atom, greater will be the force of the nucleus as the electron will be closer to the nucleus.
- Shielding – Less electrons and shells, smaller will *be the shielding which will in turn increa the nuclear charge.
- Size of Atom – Greater the size of the atom, the outer electron will become further away from th nucleus resulting in decreases in nuclear charge
- Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases.
- The size of the atom also increases down the group which makes the nuclear charge weaker
- The electron shells also increases which decreases the effective nuclear charge on the incoming electron.
- Due to all these factors, the nuclear charge decreases which decreases the tendency of gaining electrons down the group of halogen making them less reactive.
COMPARISON BETWEEN GROUP 1 and GROUP 7
| GROUP 1 Alkali Metals | GROUP 7 Halogens |
| a) Have one electron in their outermost shells | Have seven electrons in their outermost shell |
| b) They are metals | They are non metals |
| c) They react by loosing electrons | They react by gaining electrons |
| d) They form +1 ions | They form -1 ions |
| e) Their reactivitiy increases down the group | Their reactivity decreases down the group |
| f) Reactivity depends on tendency to loose an electron | Reactivity depends on tendency to gain an electron |
TRANSITION METALS
- Found between group 2 and group 3
- They are hard
- They are strong
- They are malleable and ductile
- They have higher densities than group 1 and group 2 hence they are used in construction purpose like iron.
- They show variable oxidation states
- They are used commercially as catalyst
- They form coloured compounds.
- They show the reaction with oxygen, water and halogen like group 1 but they react much slowly than alkali metals.
 Banner 7
BASIC TERMS OF PERIODIC TABLE
Periodic Table – A table that shows arrangement of all the known elements in the order of increasing atomic number. The table is organised into periods and groups.
Metals- Elements found to the left of the periodic table which are soft, shinny, conductors malleable and ductile eg Group 1; group 2 and group 3 elements
Non Metals — Elements found to the right of the periodic table which are dull, insulators Group 4,516 and 7 are non metals.
Halogens Group 7 elements are halogens as they are salt forming.
Alkali Metals Group 1 elements which react with water to form alkali
Noble Gases — Group 0 elements which are stable and do not react as they have complete outer shell
Elements found between group 2 and group 3 which have high densities, show variable oxidation states
Transition Metals and form coloured compounds
Periods- Horizontal rows of the periodic table
Groups – Vertical columns of the periodic table
Group Number – Indicates the number of electrons in the outermost shell.
Alkali Bases that are soluble in water
Displacement Reaction — When a more reactive element displaces the less reactive element from its salt
Banner 8
Q1 Look at the periodic table and give two examples of each
Metal — Li, Na
Non Metal – O2, F2
Alkali Metal – K, Li
Halogens – F, Cl
Noble Gas – He, Ar
Semi metal or metalloid – Si
Metals that form +1 ions – Li, K
Non metal that form -1 ions – F, Cl
Metal that form +2 ions – Mg, Ca
Transition metal – Fe, Cu
Q2 Write the name of most reactive halogen and most reactive alkali metals
Halogen –F Alkali Metal – Fr
Q3 Why the alkali metals gets more reactive down the group
Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus. As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge. Therefore, the tendency of atom to loose an electron increases down the group resulting in increase in reactivity down the group.
Q4 Why halogens get less reactive down the group
Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases. The size of the atom also increases down the group which makes the nuclear charge weaker. The electron shells also increases which decreases the effective nuclear charge on the incoming electron. Due to all these factors, the nuclear charge decreases which decreeases the tendency of gaining electrons down the group of halogen making them less reactive.
Q5 Write the balanced chemical equation with state symbols of
Banner 7
BASIC TERMS OF PERIODIC TABLE
Periodic Table – A table that shows arrangement of all the known elements in the order of increasing atomic number. The table is organised into periods and groups.
Metals- Elements found to the left of the periodic table which are soft, shinny, conductors malleable and ductile eg Group 1; group 2 and group 3 elements
Non Metals — Elements found to the right of the periodic table which are dull, insulators Group 4,516 and 7 are non metals.
Halogens Group 7 elements are halogens as they are salt forming.
Alkali Metals Group 1 elements which react with water to form alkali
Noble Gases — Group 0 elements which are stable and do not react as they have complete outer shell
Elements found between group 2 and group 3 which have high densities, show variable oxidation states
Transition Metals and form coloured compounds
Periods- Horizontal rows of the periodic table
Groups – Vertical columns of the periodic table
Group Number – Indicates the number of electrons in the outermost shell.
Alkali Bases that are soluble in water
Displacement Reaction — When a more reactive element displaces the less reactive element from its salt
Banner 8
Q1 Look at the periodic table and give two examples of each
Metal — Li, Na
Non Metal – O2, F2
Alkali Metal – K, Li
Halogens – F, Cl
Noble Gas – He, Ar
Semi metal or metalloid – Si
Metals that form +1 ions – Li, K
Non metal that form -1 ions – F, Cl
Metal that form +2 ions – Mg, Ca
Transition metal – Fe, Cu
Q2 Write the name of most reactive halogen and most reactive alkali metals
Halogen –F Alkali Metal – Fr
Q3 Why the alkali metals gets more reactive down the group
Down the group the atom size increases due to increase in number of electron shells. This results in the outer electron being further away from the nucleus. As the outer electron becomes further away from the nucleus the nuclear charge decreases. Increase in number of shells also increases the shielding and shields the outer electron from the nuclear charge. Therefore, the tendency of atom to loose an electron increases down the group resulting in increase in reactivity down the group.
Q4 Why halogens get less reactive down the group
Down the group the atom size increases due to increase in number of electron shells. As a result the nuclear charge decreases. The size of the atom also increases down the group which makes the nuclear charge weaker. The electron shells also increases which decreases the effective nuclear charge on the incoming electron. Due to all these factors, the nuclear charge decreases which decreeases the tendency of gaining electrons down the group of halogen making them less reactive.
Q5 Write the balanced chemical equation with state symbols of
- a) Potassium with water
- b) Lithium with oxygen
- c) Sodium with bromine
- d) Chlorine with hydrogen
Disclaimer:
I have tried my level best to cover the maximum of your specification. But this is not the alternative to the textbook. You should cover the specification or the textbook thoroughly. This is the quick revision to help you cover the gist of everything. In case you spot any errors then do let us know and we will rectify it. References: BBC Bitesize Wikipedia Wikimedia Commons Image Source: Wikipedia Wikimedia Commons Flickr PixabayMake sure you have watched the above videos and are familiar with the key definations before trying these questions. It is also good to time yourself while doing these questions so that you can work on the speed as well.
Chemistry Of The Periodic Table
- Periodic Table 1 MS
- Periodic Table 1 QP
- Periodic Table 2 MS
- Periodic Table 2 QP
- Periodic Table 3 MS
- Periodic Table 3 QP
- Properties of Transition Metals 1 MS
- Properties of Transition Metals 1 QP
- Properties of Transition Metals 2 MS
- Properties of Transition Metals 2 QP
- Properties of Transition Metals 3 MS
- Properties of Transition Metals 3 QP
